The "Dial Tone" Trap: Why eTMF Services Are Commoditizing
For the last decade, the "gold rush" in Life Sciences IT was implementing Veeva Vault eTMF (electronic Trial Master File). It was a massive digital transformation wave as pharma moved from paper to cloud. However, the data from 2025 is clear: the wave has crested. 100% of the top 20 biopharma companies and over 450 organizations globally have now standardized on Vault eTMF.
For Veeva partners, this saturation signals a dangerous pivot point. eTMF implementation has become the "dial tone" of the industry—essential, but priced as a commodity. We are seeing bill rates for generic "Vault Platform" consultants compress by 15-20% as global systems integrators (GSIs) flood the market with certified resources. If your firm’s primary revenue stream is migrating documents into eTMF or performing basic configuration updates, you are effectively trading at a "Staff Augmentation" multiple (6-8x EBITDA). The strategic value—and the premium valuation—has moved elsewhere.
The Rise of the "Unified" Clinical Platform
While eTMF is saturated, the broader Clinical Operations landscape is fracturing and expanding. Sponsors are no longer just looking for a place to store documents; they are desperate for systems that manage the complex data of decentralized clinical trials (DCTs). This is where the "Unified Clinical" opportunity lies: integrating eTMF with Vault CTMS (Clinical Trial Management System) and, most critically, Vault CDMS (Clinical Data Management System).
The CDMS Multiplier: From Document Manager to Data Strategist
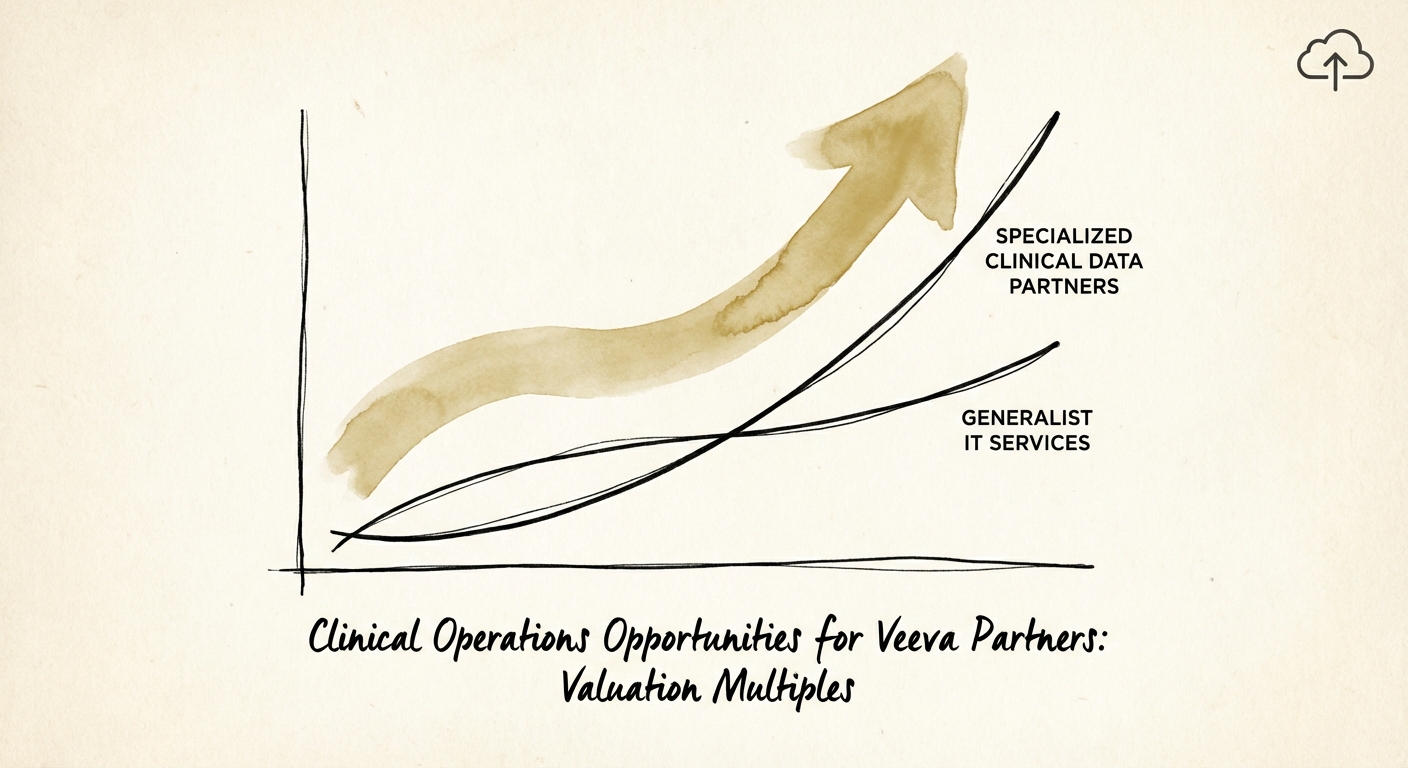
The valuation gap between "Document Shops" (eTMF focus) and "Data Strategists" (CDMS/CTMS focus) is widening to historic levels. In 2025, private equity deal value in Healthcare IT doubled to $32 billion, driven largely by assets that can handle proprietary clinical data, not just files. Partners who have mastered Vault EDC (Electronic Data Capture) and CDMS are commanding multiples of 12x to 14x EBITDA.
Why the premium? Complexity acts as a defensive moat. Migrating a clinical trial’s data (patient forms, lab results, safety signals) is exponentially more complex and higher-risk than migrating its documents (protocols, investigator brochures). A failed document migration is a compliance headache; a failed data migration is a clinical failure that delays drug approval. Partners who can execute these high-stakes CDMS initiatives become "sticky" strategic advisors, not just interchangeable vendors.
The Agentic AI Frontier
The next phase of value creation is Veeva AI. With the release of Agentic AI tools embedded in Vault, sponsors need partners who can design workflows where AI agents handle routine TMF classification or data reconciliation. Partners who position themselves as "AI Implementation Leaders"—specifically in reducing clinical cycle times—are seeing a distinct "Innovation Premium" in due diligence. The market is paying for the capability to accelerate trials, not just administer them.
The Diagnostic: Are You a Commodity or a Strategic Asset?
To determine if your Veeva practice is positioned for a premium exit, evaluate your revenue mix against these three benchmarks:
- Revenue Composition: If >70% of your revenue is derived from eTMF implementation and managed services, you are in the "Commodity Zone." A premium partner should have at least 40% of revenue coming from complex modules like CTMS, CDMS, or Study Startup.
- Talent Density: Do your consultants mostly hold "Vault Platform" associate certifications, or do you have deep density in "Vault Clinical Data Management" and "Vault CTMS"? The former is easy to hire; the latter is a scarce asset that acquirers pay for.
- Outcome-Based Pricing: Are you billing by the hour for configuration, or by the "Study Start-Up"? Premium firms are moving to managed service models where they own specific clinical outcomes (e.g., "Site Activation in 30 Days"), decoupling revenue from headcount.
The window to pivot is narrowing. As Veeva migrates its entire CRM base to Vault CRM by 2030, the ecosystem will be flooded with "generalist" work. The partners who win big in 2026 will be those who have already staked their claim in the complex, high-stakes world of Clinical Data.