The Great Bifurcation: Commercial Red Oceans vs. Quality Blue Oceans
For the last decade, the "Veeva Playbook" for private equity was simple: acquire a Salesforce-based CRM consultancy, pivot them into the booming Life Sciences vertical, and ride the Veeva Commercial Cloud wave. That wave has crested.
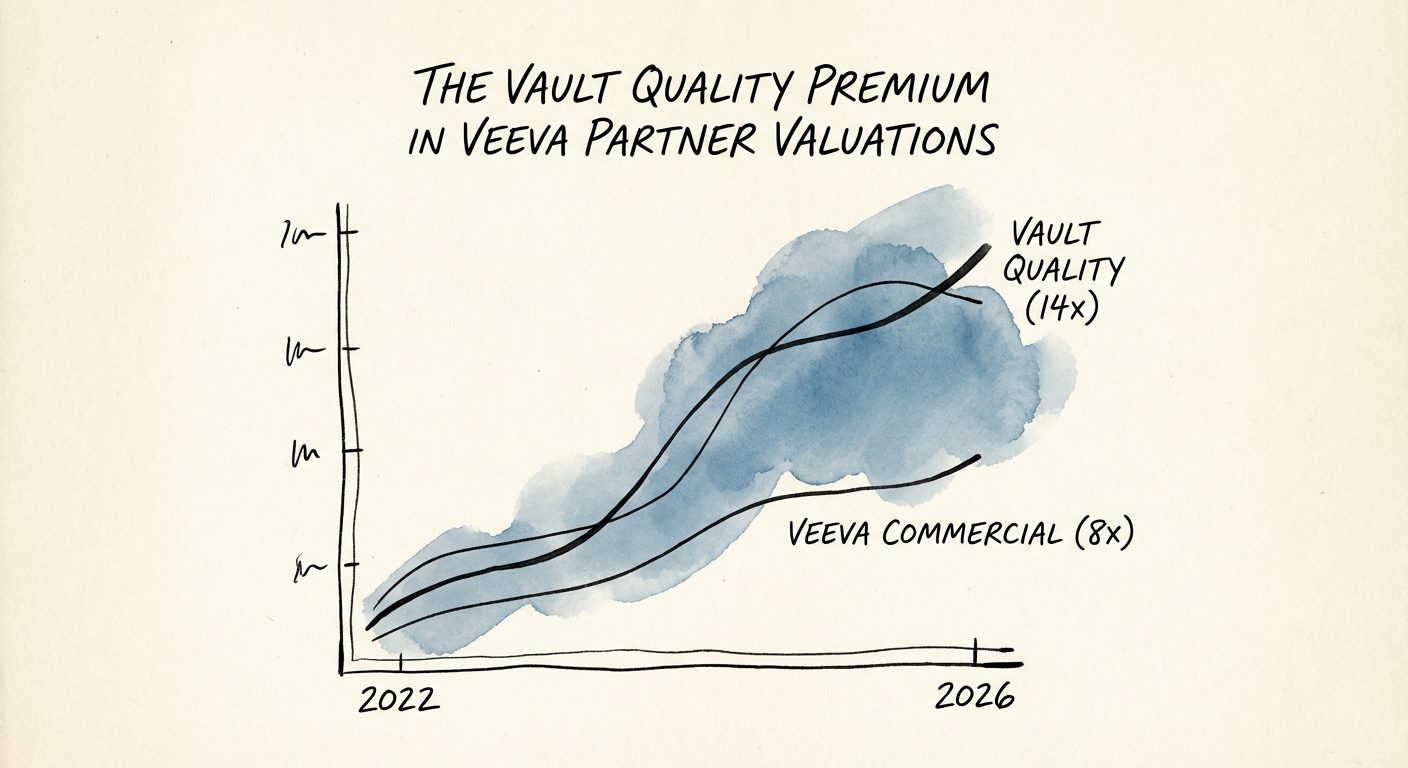
Today, the market for Veeva Commercial services—focused on CRM, PromoMats, and sales enablement—has commoditized. With Veeva CRM migrating off Salesforce onto the proprietary Vault platform by 2030, generalist CRM shops are facing an existential skills gap. Consequently, valuation multiples for "Commercial-first" Veeva partners have compressed to the 8x-10x EBITDA range, mirroring broader IT services trends.
However, a second, more lucrative market has emerged within the same ecosystem: Vault Quality. Partners specializing in the "Development Cloud" (Quality, Regulatory, and Clinical) are trading at a significant premium, often commanding 12x-15x EBITDA. Why? Because while Commercial spend is discretionary (driven by sales cycles), Quality spend is regulatory (driven by the FDA). In a macroeconomic downturn, pharma companies fire sales reps, but they cannot defund their Quality Management Systems (QMS) without risking a Warning Letter or a Consent Decree.
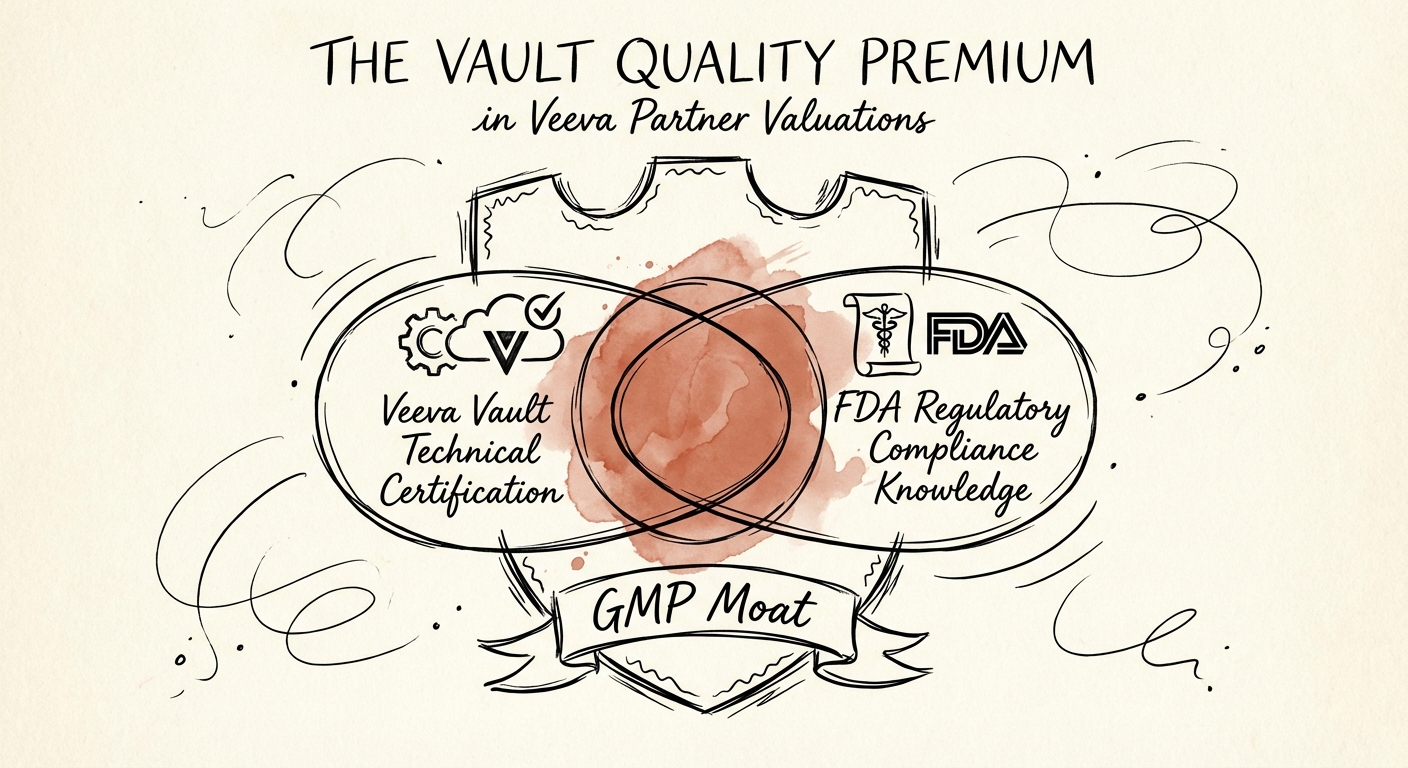
The "GMP Moat": Why Generalists Can't Cross the Chasm
The valuation premium for Vault Quality shops isn't just about software; it's about the scarcity of the talent. Implementing Veeva QualityDocs or QMS requires consultants who understand Good Manufacturing Practice (GMP), 21 CFR Part 11 compliance, and complex validation workflows. A developer who knows Apex code can't simply "figure out" a Corrective and Preventive Action (CAPA) workflow; the regulatory risk is too high.
The Rate Resilience Index
This "GMP Moat" creates distinct unit economics. While Commercial implementation bill rates have softened to $150-$175/hr due to offshore competition, specialized Quality consultants consistently command $195-$250/hr. Furthermore, the "stickiness" of these engagements is superior. Once a Quality partner validates a system, they effectively own the "compliance chain of custody," leading to multi-year managed services contracts with 90%+ retention rates.
For PE buyers, this creates a defensive asset profile. A Commercial partner's revenue fluctuates with drug launches. A Quality partner's revenue is anchored to the license to operate. As healthcare IT due diligence increasingly focuses on compliance risks, these specialized firms offer a "safe harbor" with growth upside.
The Vault CRM Migration Opportunity
The irony of the current market is that the "Commercial" partners are now scrambling to acquire "Quality" DNA. With Veeva moving its entire CRM user base (80% market share) to the Vault platform, the underlying technology stack for Commercial is becoming the same as Quality. This has triggered a wave of consolidation.
Smart acquirers are buying Vault Quality firms not just for their stable cash flows, but as the "Trojan Horse" for the next decade of Commercial work. A partner that understands the Vault object model (the architecture behind Quality) is infinitely better positioned to handle the massive Vault CRM migrations beginning in 2025 than a legacy Salesforce shop. This convergence is driving the 14x multiples we see for "Vault-Native" firms.
Investors should look for three specific indicators of a "Premium" Veeva asset: 1) A ratio of Development Cloud revenue >50% (vs. Commercial), 2) A "Managed Validation" recurring revenue stream, and 3) Referenceable expertise in "Unified" implementations (e.g., connecting QualityDocs with Regulatory Information Management). These are the assets that will command the migration era premiums.