The Life Sciences Premium: Why Veeva Partners Outperform Generalists
In the hierarchy of IT services valuations, Veeva partners occupy a rare tier. While generalist Salesforce or Microsoft systems integrators typically trade at 6x-8x EBITDA, specialized Life Sciences partners are commanding 12x-14x multiples in 2025/2026. This "Life Sciences Premium" is driven by a defensive moat that generalists cannot easily cross: the high barrier of GxP compliance, validation, and regulatory expertise.
However, not all Veeva partners share in this premium. A dangerous bifurcation has emerged in the ecosystem between "Commercial Cloud" legacy partners and "Vault" platform strategists. With Veeva's strategic shift away from Salesforce technology (completing the migration to Vault CRM by 2030), the valuation gap is widening.
The "Vault Catalyst" for 2026
The migration to Vault CRM is creating a massive, one-time services tailwind. Partners who have mastered the Vault platform—encompassing Clinical, Quality, Regulatory, and now CRM—are seeing bill rates 35% higher than their Commercial-only counterparts. Acquirers are no longer looking for "implementation capacity"; they are buying "migration assurance." Firms that can navigate the technical and data complexities of moving a global pharma commercial operation from Salesforce to Vault are trading at premium valuations, while those stuck in legacy support models are seeing their multiples compress.
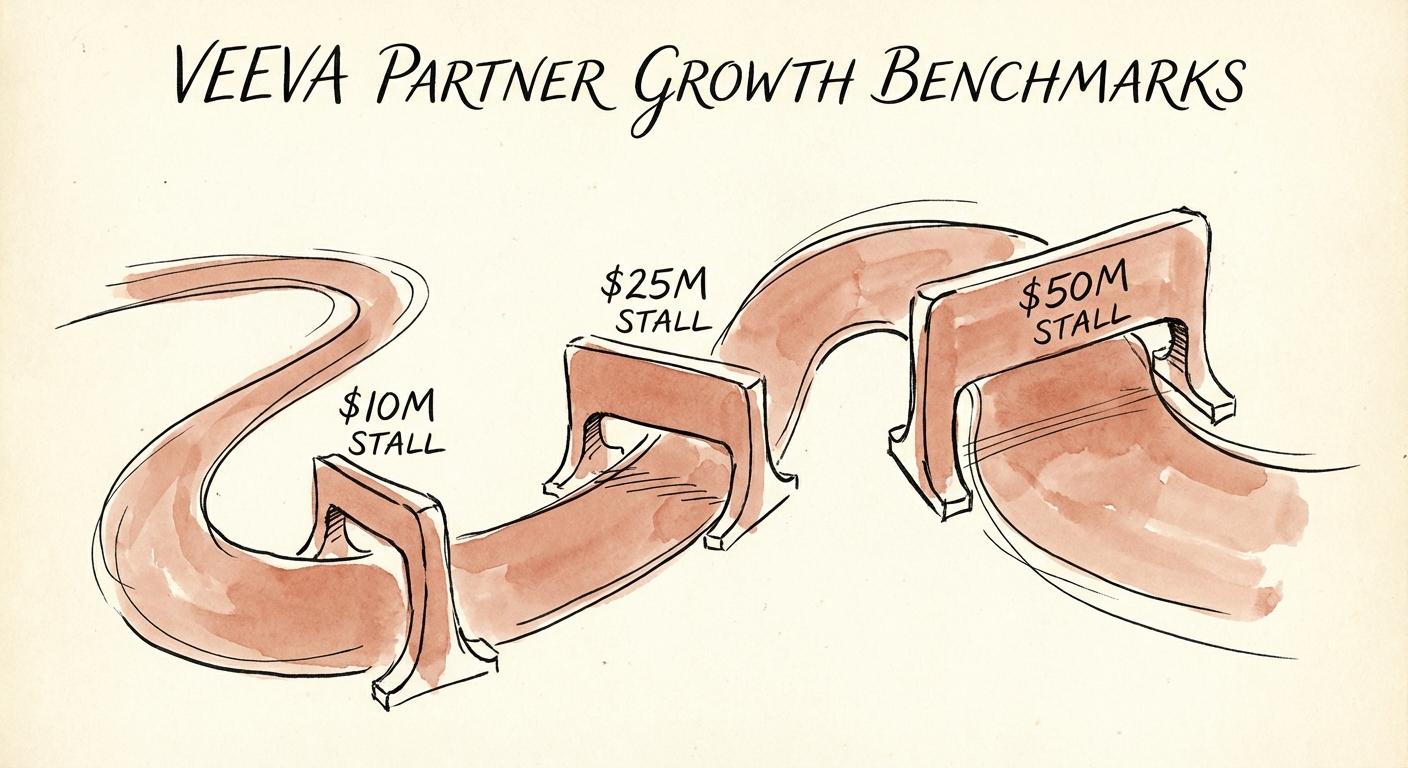
The Three Growth Stalls: Where Veeva Partners Get Stuck
Scaling a Veeva practice requires navigating distinct "valleys of death" where revenue growth often stalls due to operational complexity or lack of specialization. Our data from 50+ Life Sciences services firms reveals three critical inflection points.
Stall 1: The $10M "Commercial Only" Ceiling
At $5M-$10M in revenue, many partners rely heavily on Commercial Cloud implementations (CRM, PromoMats). While this market is large, it is commoditized. These firms often struggle to break into the more lucrative R&D side of the house. Customer concentration is high, often with 1-2 major pharma clients driving 60% of revenue.
Stall 2: The $25M "R&D Chasm"
To break past $20M, a partner must diversify into R&D Cloud (Clinical, Quality, Regulatory). This requires a different talent breed—consultants who understand TMF (Trial Master File) structures and QMS (Quality Management Systems), not just sales workflows. Partners who fail to cross this chasm often see utilization drop as they try to retrain sales-focused delivery teams for highly technical regulatory projects.
Stall 3: The $50M "Global Delivery" Hurdle
At $50M, the game changes from "expert delivery" to "managed services at scale." Global pharma clients demand follow-the-sun support for their critical compliance systems. Partners who cannot establish a nearshore/offshore delivery center (often in Eastern Europe or India) with GxP-compliant security standards will lose Application Managed Services (AMS) contracts to the Global Systems Integrators (GSIs). Without this recurring revenue engine, valuation multiples cap out at 8x.
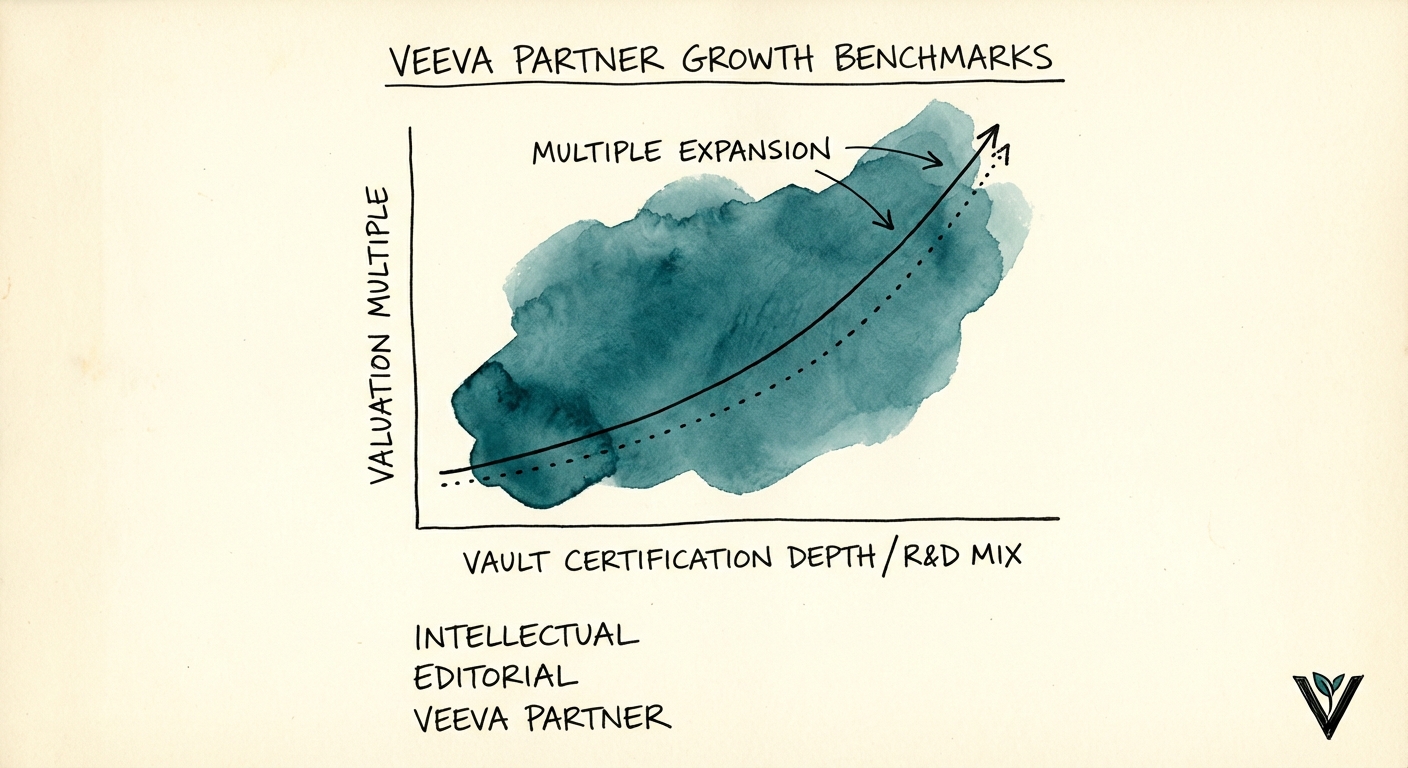
Valuation Drivers: The Metrics That Matter
For PE sponsors and founders evaluating exit readiness, four metrics differentiate a 6x asset from a 14x strategic platform.
- Vault Certification Depth: Total certifications are vanity metrics. The ratio of "Vault Platform" and "R&D" certifications to total headcount is the quality metric. Best-in-class firms maintain a 1:3 ratio of R&D to Commercial consultants.
- Blended Bill Rate: While generalist SI bill rates hover around $175/hr, premium Veeva Vault architects command $225-$275/hr. If your blended rate is below $190, you are likely stuck in low-end staff augmentation.
- Revenue Mix (Project vs. AMS): The highest valuations go to firms with 30%+ revenue from recurring Managed Services. Pure project-based firms are viewed as risky "revenue treadmills."
- Utilization Hygiene: Unlike the 85% "burnout" targets of generic shops, elite Veeva consultancies optimize for 72% utilization, allowing time for the constant up-skilling required by Veeva's rapid release cycle (3x/year).